articles in 2026
all / 2026 / 2025 / 2024 / 2023 / 2022 / 2021 / 2020 / 2019 / 2018 / 2017 / 2016 / 2015 / 2014 / 2013 / 2012 / 2011 / 2010 / 2009 / 2008 / 2007 / 2006 / 2005 / 2004 / 2003 / 2002 / 2001 / 2000 / 1999 / 1998 / 1997 / 1996 / 1995 / 1994 / 1993
-
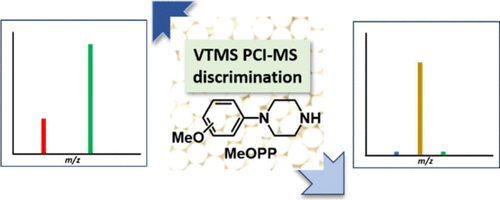 Vinyltrimethylsilane Chemical Ionization Tandem Mass Spectrometry Separates Methoxyphenylpiperazine IsomersZiaho Qin, Shinji Tsunoi*, Koya Hachiri, Ikuya Shibata, and Tadashi Mori*Anal. Chem., 2026, in press.
Vinyltrimethylsilane Chemical Ionization Tandem Mass Spectrometry Separates Methoxyphenylpiperazine IsomersZiaho Qin, Shinji Tsunoi*, Koya Hachiri, Ikuya Shibata, and Tadashi Mori*Anal. Chem., 2026, in press.Novel synthetic drugs often contain aromatic positional isomers that are difficult to distinguish by conventional mass spectrometry due to similar fragmentation patterns and retention times. Isomers of methoxyphenylpiperazine (MeOPPs) exemplify this analytical challenge. Here, we employed chemical ionization tandem mass spectrometry using vinyltrimethylsilane as the reagent gas to analyze trifluoroacetyl-derivatized MeOPP isomers. This approach generated diagnostic fragment ions unique to the ortho, meta, and para isomers, enabling their unambiguous discrimination in a single analytical run. Density functional theory calculations supported the proposed fragmentation pathways and provided mechanistic insight into the observed isomer-specific selectivity. This straightforward and robust method offers a promising strategy for precise isomer identification in forensic and pharmaceutical contexts where the accurate differentiation of synthetic drug isomers is critical.
@article{qin2026vinyltrimethylsilane, title = {Vinyltrimethylsilane Chemical Ionization Tandem Mass Spectrometry Separates Methoxyphenylpiperazine Isomers}, author = {Qin, Ziaho and Tsunoi, Shinji and Hachiri, Koya and Shibata, Ikuya and Mori, Tadashi}, journal = {Anal. Chem.}, pages = {in press}, year = {2026}, month = mar, publisher = {ACS Publications}, doi = {10.1021/acs.analchem.5c08125}, url = {https://doi.org/10.1021/acs.analchem.5c08125}, dimensions = {true}, tab = {paper}, } -
 Small molecule helical emittersTadashi Mori*Chem. Soc. Rev., 2026, 55, 1999–2023.
Small molecule helical emittersTadashi Mori*Chem. Soc. Rev., 2026, 55, 1999–2023.The development of materials exhibiting circularly polarized luminescence (CPL) is a key area of research for next-generation optical technologies, including 3D displays and secure communications. The central goal in this field is to create chiral emitters with a high luminescence dissymmetry (gCPL) factor, a measure of the emission’s chirality. While theoretically reaching ±2, practical values in small organic molecules have historically been much lower, on the order of 0.001 or less. This summary outlines the core strategies in molecular design focusing on helical emitters that have recently enabled significant breakthroughs, pushing g values beyond the 0.01 threshold. The magnitude of g factor is determined by the cosine of the angle between the electric (μe) and magnetic (μm) dipole transition moments, as well as their respective magnitudes. Consequently, the most successful research has moved beyond simple screening and has focused on rationally engineering molecules to optimize this relationship. One of the most direct strategies has been to design rigid, helical molecules where high symmetry forces the μe and μm to be parallel. By enforcing D2 and other symmetry in certain helicenes, helical nanographenes and related structures, researchers have minimized the angle between the moments, thus maximizing the cosine term and leading to a significant enhancement in the g factor value. A second, distinct approach targets the magnitude of the μm. In most organic chromophores, μm is inherently small, limiting the potential g factor intensity. To overcome this, researchers have designed for example belt-shaped macrocyclic molecules that function as molecular-scale solenoids. The cyclic arrangement of chromophores induces a large, circulating electric current in the excited state, which in turn generates a powerful μm along the cylinder’s axis. A third innovative strategy circumvents the limitation of a small intrinsic μm by leveraging exciton coupling between two and more chromophores. In these systems, two π-conjugated units such as pyrene are held in a fixed, chiral arrangement. Upon photoexcitation, they form an intramolecular excimer, a transient excited-state complex with a well-defined helical geometry. The resulting CPL signal originates from the chiral interaction of the two strong electric transition moments, generating a large rotational strength and a high g factor without relying on the weak magnetic moment of the individual units. The progress in CPL-active materials is a testament to the power of targeted molecular engineering. As seen in the state-of-the-art examples in the review, the field has matured to a point where the fundamental photophysical principles governing CPL are being directly translated into synthetic molecular designs. While current high-performing materials are often complex and synthetically challenging, these proof-of-concept molecules validate the core design strategies.
@article{mori2026small, title = {Small molecule helical emitters}, author = {Mori, Tadashi}, journal = {Chem. Soc. Rev.}, pages = {1999--2023}, year = {2026}, volume = {55}, issue = {4}, publisher = {Royal Society of Chemistry}, doi = {10.1039/d5cs01270h}, url = {https://doi.org/10.1039/d5cs01270h}, dimensions = {true}, tab = {review}, }